Victoria Gray was given life expectancy of seven years after being diagnosed with sickle cell disease (SCD) at three months.
For 34 years, she had lived with sickle cell disease. This genetic condition twisted her normally round red blood cells into sharp, crescent shaped ones. The abnormal cells clogged her blood vessels and triggered excruciating pain crises. Growing up with it was difficult, never knowing when the next crisis will come. By 2019, she was hospitalized almost monthly, sometimes for weeks at a time. Her young children watched their mother disappear into a cycle of pills, transfusions, and hospital stays that offered temporary relief but no hope of cure.
“I had basically given up,” Victoria recalls. “The doctors could manage my pain, give me blood transfusions, prescribe medications. But none of that was going to change the fact that my DNA was programmed to make defective blood cells. You can’t take a pill to fix your genes.”
Traditional medicine had reached its limits. A single letter change in her genetic code that caused a lifetime of suffering. No amount of conventional treatment could rewrite that fundamental programming error.
While exploring an option of a bone marrow transplant in Nashville, she found a lifeline. It came in the form of a clinical trial for SCD using gene-editing technology. Victoria became one of the first patients in the world to receive a groundbreaking treatment. Doctors collected her hemopoietic stem cells from blood. They used molecular “scissors” called CRISPR to edit her DNA. Then, they returned the corrected cells to her body.
This was in 2019, since then Victoria has not experienced a pain crisis. Her children have their mother back, and Victoria is planning a future she never thought possible.
Welcome to the age of genetic medicine, where we are no longer limited to treating symptoms. We can rewrite the code of life itself.
The CRISPR Revolution Explained
CRISPR-Cas9, often called “molecular scissors,” represents the most precise editing tool biology has ever created. Unlike traditional medications that temporarily alter how cells function, CRISPR makes permanent changes to DNA.
How Traditional Treatment Failed
For diseases like sickle cell, conventional medicine was trapped in an endless cycle of damage control, treating symptoms only. Sickle cell disease occurs when patients inherit two copies of a mutated gene that produces abnormal hemoglobin. This faulty hemoglobin causes red blood cells to become rigid and sickle-shaped, blocking blood flow and causing pain, organ damage, and shortened lifespans.
Traditional treatments included:
- Pain medications during crises (treating symptoms, not causes)
- Blood transfusions to replace sickled cells (temporary fix requiring lifelong repetition)
- Hydroxyurea to reduce sickling frequency (helpful but not curative)
- Bone marrow transplants (curative but requiring matched donors and carrying high risks)
The CRISPR Breakthrough
CRISPR works by exploiting a cellular editing system that bacteria evolved to fight viruses. Scientists program guide RNAs to locate specific DNA sequences, then use Cas9 proteins to make precise cuts. But here is the revolutionary part: instead of trying to fix the sickle cell mutation directly, CRISPR treatments like Casgevy use a clever workaround.
Every person carries a “backup” hemoglobin gene called fetal hemoglobin that normally shuts off after birth. CRISPR edits the genetic switch that turns off fetal hemoglobin. It essentially reactivates a healthy hemoglobin program that was already in the patient’s cells. As a result patients start producing normal, round red blood cells again.
Beyond Repair to Restoration
This represents a fundamental shift in medical thinking. Instead of managing a genetic disease, CRISPR can eliminate it. Instead of working around faulty genes, it can reactivate dormant healthy ones. Instead of lifelong treatment cycles, it offers the possibility of one-time cures.
Real Patients, Real Results
The numbers from CRISPR clinical trials read like medical miracles, but behind each statistic is a transformed life.
Victoria Gray: From Hospital Bed to Normal Life
Victoria’s transformation began in July 2019 when she entered a clinical trial at Sarah Cannon Research Institute. The process was not simple or easy for her. Her own stem cells were collected over three days for six hours a day. Then while the cells were modified, Victoria underwent a chemotherapy treatment to prepare her body for the infusion of modified cells. Finally, the cells she received cells back by an IV infusion.
The results exceeded everyone’s expectations. Within months, Victoria’s fetal hemoglobin levels soared from nearly undetectable to over 40% of her total hemoglobin. Her sickled cells virtually disappeared. More importantly, her pain crises stopped entirely.
“I can contribute to my household and show up for my family,” Gray said. “I feel like I’m a part of society now. I had been isolated for years, but now I’m free to be me and dream again. I don’t have to give up my dreams because of pain anymore.” (AABB News: An Answered Prayer: Warrior Reflects on Her Experience as the First Patient Treated With CRISPR for Sickle Cell Disease)
Expanding Success Stories
Victoria is not the only patient to receive this life saving treatment. The clinical trial included 44 patients, 31 of them were followed for at least 18 months post-treatment. Out of those the treatment was successful in 29 patients. Clinical trials of Casgevy (exagamglogene autotemcel) have shown remarkable consistency:
- 95% of sickle cell patients experienced no pain crises for at least 12 months after treatment
- 100% of beta thalassemia patients became transfusion-free or achieved significant transfusion reduction
- No serious treatment-related side effects in long-term follow-up
The trials included patients like:
- Jimi, sickle cell disease patient got his treatment at the age of 35. Within two weeks his pain was gone and his life was completely transformed.
- Johnny, a teenager with sickle cell disease, got his treatment in October 2019. He described the process as cool and freaky but has been doing well since the treatment. He now plays drums, something that would have not been possible before.
- Jayden, a teenager with sickle cell beta thalassemia got therapy after the approval by the FDA. His parents are optimistic about his future and freedom from pain and endless hospital stays.
Long-term Monitoring
The longest-treated patients are now over four years post-CRISPR therapy, with maintained benefits and no late-onset complications. Researchers continue monitoring for long-term safety, but early results suggest the genetic changes remain stable and beneficial.
These aren’t just medical statistics. They represent fundamental changes in life trajectory. Patients who expected shortened lifespans filled with medical interventions are now planning decades-long futures with minimal medical care.
Beyond Sickle Cell Disease
While sickle cell disease and beta thalassemia represent CRISPR’s first major victories, the technology’s potential extends far beyond blood disorders. Researchers are applying CRISPR’s precision to tackle some of medicine’s most challenging genetic conditions.
The Personalized Medicine Breakthrough
Perhaps most remarkably, researchers at Children’s Hospital of Philadelphia recently treated an infant with a completely personalized CRISPR therapy. The baby had a rare metabolic disorder called carbamoyl phosphate synthetase 1 deficiency. It typically has poor prognosis, improved only by very early optimal treatment.
Scientists designed a custom CRISPR treatment specifically for this child’s genetic mutation. Treatment was manufactured it in just weeks, and successfully corrected the defective gene. The child is now developing normally. The first person in the world to receive a fully personalized genetic medicine designed from scratch.
The Expanding Pipeline
Leber Congenital Amaurosis 10 (LCA10) treatment was to be the first one to deliver CRISPR therapy directly into patient’s eyes. While early trial results were promising the EDIT-101 trial was stopped. The efficacy was below expectations with only three of eleven participants showed meaningful improvements. This failure might have resulted from a difference in delivery method or other unknown reason. However, this has not stopped new clinical trials using CRISPR technology. Current CRISPR clinical trials target among others the following conditions:
- Hereditary Transthyretin (ATTR) Amyloidosis, NTLA-2001 (CRISPR-Cas9 in vivo) by Intellia Therapeutics
- Hereditary Angioedema (HAE), NTLA-2002 – In vivo CRISPR-Cas9; therapy candidate that is designed to disrupt the target gene kallikrein B1 (KLKB1) in hepatocytes; moving toward global Phase 3
- cancer immunotherapy, targeting T cells, specifically editing the T cell receptor for potential treatment of breast, colon and lung cancer.
- Various blood cancers
The ultimate promise lies in fully customized treatments. The infant metabolic disorder case demonstrates this potential. CRISPR platforms could theoretically create bespoke genetic medicines for any patient and any genetic condition. This would allow the shift from the blockbuster drugs that help millions to precision medicines that cure individuals.
The Reality Check
CRISPR represents genuine medical revolution, but it’s important to understand both its promise and current limitations.
What’s Available Now
As of 2024, patients can access CRISPR treatment through:
- Casgevy: FDA and EMA-approved for sickle cell disease and beta thalassemia in patients 12 and older
- Clinical trials: Multiple ongoing studies for other genetic conditions
- Specialized treatment centers: Limited to major academic medical centers with gene therapy capabilities
The Process Reality
CRISPR treatment isn’t as simple as taking a pill. The current process for Casgevy includes:
- Extensive screening and genetic testing
- Cell collection procedures – a long procedure, which can extend into multiple days until the adequate number of cells are collected
- Hospital stay for chemotherapy conditioning, which comes with its own risks and challenges for patients
- Cell infusion and recovery period
- Long-term monitoring for safety and effectiveness
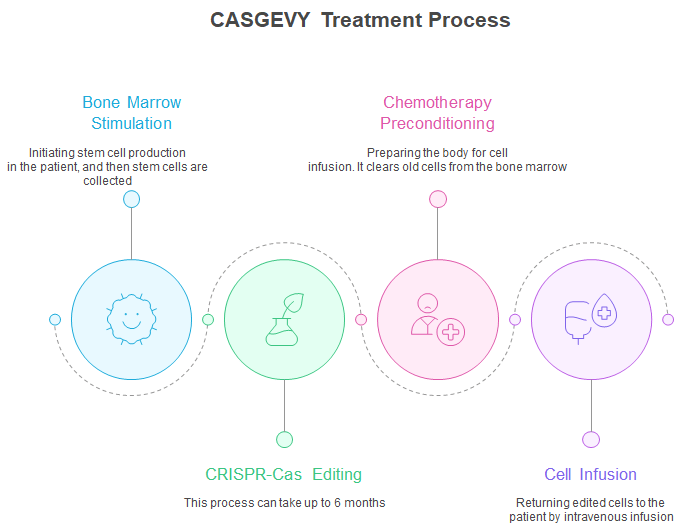
The entire process takes several months and requires treatment at specialized centers. While current results are overwhelmingly positive, CRISPR therapy requires lifelong monitoring. Researchers continue studying:
- Effects on fertility and inheritance (though current evidence suggests edited blood cells don’t affect reproductive cells)
- Long-term genetic stability of edited cells
- Potential off-target genetic changes
- Interactions with other medical conditions or treatments
Cost and Access Considerations and Challenges
Casgevy costs $2.2 million per patient, raising important questions about access and affordability. However, economic analyses suggest this one-time cost may be offset by eliminating lifelong medical expenses. Traditional sickle cell disease care costs approximately $1.6 million over a patient’s lifetime, not including reduced quality of life and shortened lifespan. Sickle cell disease affects about 1000 babies a year and estimated 20 million globally. so while there are a lot of potential patients, there are challenges with the access to the new treatment. The two major issues with this novel treatment:
- The therapy is is limited to major medical centers, creating geographic and socioeconomic barriers to access. Efforts are underway to expand treatment availability and develop more accessible delivery methods.
- The current price tag of USD 2.2 M to 3.1 M for the treatment puts it out of reach of those who need it the most. This includes a lot of less affluent people in the US and in countries of the sub-Saharan Africa. It is also important to remember that people with sickle cell anemia cannot work full time due to ongoing issues.
In the US, many of those needing the treatment are covered by Medicaid. However, with the latest cuts to Medicaid programs there could be challenges to getting the treatment covered.
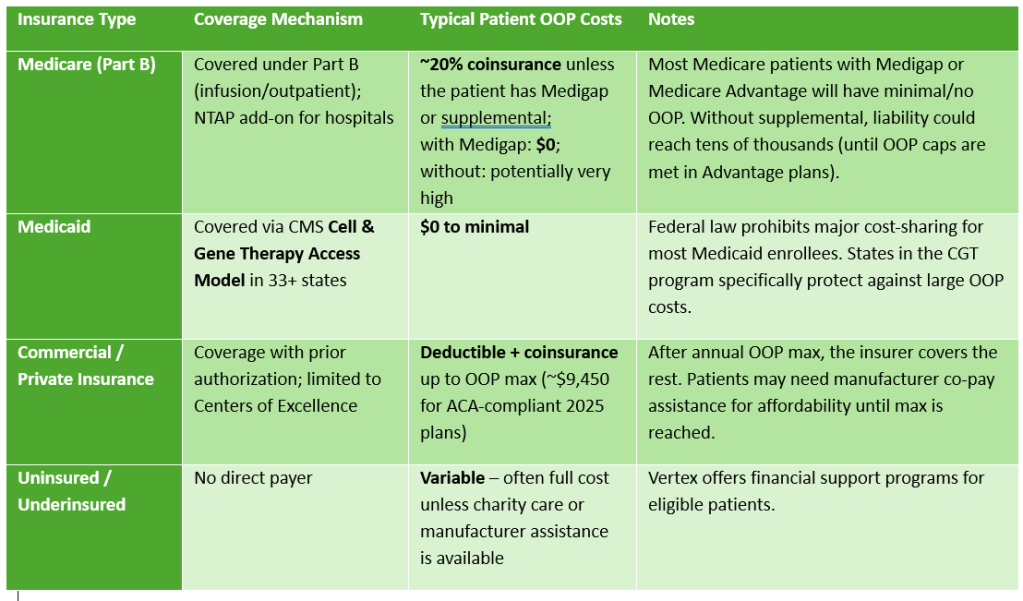
Insurance coverage is evolving. Some payers are developing innovative payment models. These models spread costs over time or tie payments to treatment outcomes.
According to Vertex, Casgevy is approved in Bahrain, Canada, the EU, Iceland, Saudi Arabia, Liechtenstein, Norway, Switzerland, United Arab Emirates, the UK and the US. We already commented on the reimbursement in the US. Canada and the UK approve public funding if certain conditions are met. In general, patients with the most severe disease will be prioritized for treatment. In the EU, there is not full transparency of the costs. However, specialized centers are to provide the treatment. Based on public information the funding is available in Austria, Germany, Luxembourg, and Italy, as well as Bahrain, and Saudi Arabia.
A New Chapter in Medicine
Victoria’s story represents the beginning of genetic medicine’s true promise. We are moving beyond the limitations of pills that mask symptoms and surgeries that repair damage. We enter an era where we can rewrite the fundamental instructions that govern health and disease.
For the estimated 400 million people worldwide living with genetic conditions, CRISPR offers something traditional medicine never could: the possibility of cure instead of management, hope instead of resignation, and normal lifespans instead of shortened ones.
A decade ago CRISPR was used mostly in the lab and was not consider a medical tool. Now we can edit patient’s genome and fix the problem. While it is promising avenue, not all diseases will be treatable with CRISPR. Also not all will use the same cell delivery system Casgevy is using. But the door to the future was opened and patients can have hope they could not before.
For more information
For more information on suitability of Casgevy as the treatment for your sickle cell anemia or beta-thalassemia talk to your doctor. You can also visit Vertex site, check news on sickle cell disease, or visit sickle cell society.
References and further reading:
- Sickle Cell Salvation: The Victoria Gray Interview, accessed 01 Sep 2025
- CRISPR gene-editing success for sickle cell raises new questions : Shots – Health News : NPR, accessed 01 Sep 2025
- CRISPR gene-editing treated her sickle cell disease and she’s still thriving : Shots – Health News : NPR, accessed 01 Sep 2025
- AABB News: An Answered Prayer: Warrior Reflects on Her Experience as the First Patient Treated With CRISPR for Sickle Cell Disease, accessed 01 Sep 2025
- CRISPR’s Breakthrough Success: Real Patient Stories and Treatment Results – Health & Wellness Canada, accessed 01 Sep 2025
- FDA approves cure for sickle cell disease, the first treatment to use gene-editing tool CRISPR, accessed 01 Sep 2025
- FDA Approves First Gene Therapies to Treat Patients with Sickle Cell Disease | FDA, accessed 01 Sep 2025
- From struggling with sickle cell disease to climbing mountains – a firsthand account of how gene therapy has been life changing, accessed 01 Sep 2025
- Opinion: The revolutionary sickle cell therapies | CNN, accessed 01 Sep 2025
- Sickle cell disease: Falmouth teen starts new human gene therapy, accessed 01 Sep 2025
- Baby saved by gene-editing therapy ‘graduates’ from hospital, goes home – ABC News, accessed 01 Sep 2025
- Details for: CASGEVY – Drug and Health Products Portal, accessed 01 Sep 2025
- Exagamglogene Autotemcel (Casgevy) | Canadian Journal of Health Technologies, accessed 01 Sep 2025
- Casgevy Cost: A Detailed Look at Pricing Factors – Biology Insights, accessed 01 Sep 2025
- NHS to offer ‘groundbreaking’ sickle cell gene therapy, accessed 01 Sep 2025
- CRISPR gene therapy Casgevy granted European approval | HealthCare Middle East & Africa Magazine, accessed 01 Sep 2025
- Gene Editing Therapy Casgevy Now Funded In 8 Countries But French Access Withdrawn, accessed 01 Sep 2025
- Casgevy: Launch sequence and price analysis of the first marketed CRISPR therapy, accessed 01 Sep 2025
- Casgevy (exagamglogene autotemcel) for sickle cell disease, accessed 01 Sep 2025


Leave a comment